Microbiology for Nursing/Allied Health (Lab)
Section outline
-
Welcome to [SUBJ ####]!
This lab course is designed for students enrolled in allied health sciences or nursing programs. Principles of microbiology, with emphasis on health and disease, include microbial diversity and physiology, host-microbe interactions, immune responses, infection prevention and control, and research procedures.Course Introduction
Upon successful completion of this course, the student will be able to:
- Distinguish between bright-field microscopy and dark-field microscopy (if fluorescent microscopes are available).
- Develop and apply a general understanding of staining techniques that will include performing: simple staining, negative staining, Gram staining, spore staining, and acid-fast staining techniques, and formulating conclusions based on the performance of activities.
- Study and apply the preparation of culture media, cultivation of anaerobic bacteria, and enumeration of bacteria using the Standard plate count.
- Identify unknown bacteria using various physiological tests.
- Demonstrate basic culture techniques and provide interpretations needed to function in the healthcare setting.
- (In a virtual setting) or (Face-to-Face setting) Perform essential microbiological techniques in a simulated environment with confidence, utilizing digital tools to analyze and interpret virtual and hands-on experimental data, and apply their virtual and/or hands-on laboratory skills to clinical and public health contexts in nursing and allied health.
Structure of the CourseAdopting institution should provide learners information on how to navigate the course. Consider adding an introductory navigation video. Text description could include, for example:Describe the course structure, such as number of modules, to allow the learner to understand how the learning process if structured and carried out.
Navigating the Course
This course is set up in Modules covering various topics which may be accessed from the course navigation menu on the left or by scrolling below. Modules may be collapsed in the menu and it the body of the course to minimize scrolling. Each module includes the relevant chapters followed by various activities, which may include discussion forums, listening activities and quizzes, practice quizzes, module tests, and other relevant activities as appropriate for each module. Many items are required and may be marked as completed automatically when the activity has been submitted (the broken check box), but others will marked as done by the student (the solid check box).Please move through the items below and continue through the Learner Support and Getting Started modules before moving on to Module 1. Be sure to check for announcements and due dates to stay on track. This course and its contents are developed by the authors: and licensed under a Creative Commons Attribution 4.0 International License by LOUIS: The Louisiana Library Network, except where otherwise noted.
This course and its contents are developed by the authors: and licensed under a Creative Commons Attribution 4.0 International License by LOUIS: The Louisiana Library Network, except where otherwise noted. Adopting instructors should edit the About Your Instructor and Office Hours Information pages in this Module.
-
Adopting instructors should edit all pages in this module to reflect their institution's policies.
-
This module contains all the items you should review and complete before you begin Module 1. Before moving on, be sure to:
- Check the News and Announcements Forum
- Read the Course Syllabus
- Introduce yourself to the class
- Read the instructions for the Q & A Forum
- Review academic integrity expectations in the course
Good luck in the course!-
This forum contains general news and announcements. You can find all announcements listed in the "Latest News" block in Course Tools within this course or on your My Courses page.
-
Google Doc Syllabus Template: Use this template to create a syllabus and attach as a Word doc.
-
Use this forum to tell us a little about yourself and your interests. Some topic ideas:
- What is your field of study/research interest or concentration?
- What are you most interested in learning about in this class and why?
- Have you ever taken an online class before?
- Any other information you would like to share with your classmates, such as special interests or activities.
Post a picture! We look forward to meeting you.
-
Use this forum to ask your instructor any questions you have about the course. You may post at any time, and your instructor will respond here. Be as specific as possible.
Please keep in mind that others can see your posts, so do not post any personal information. If you have questions about your grade, please email your instructor directly. You can expect a response to posts and emails within [X] hours. [Recommendation is 24 hours M-F, next business day on weekends.]
Subscription should be set to Auto.
-
Provide specific and contextualized information about how students can comply with institutional academic integrity policies and standards as they complete assessments in the course.
-
Use the information in this module to customize the template to your needs. This module is currently hidden from students, and available for you to refer to throughout the semester.
-

The course objectives are developed from recommendations of the American Society for Microbiology about the undergraduate curriculum for Microbiology courses and professional programs in allied health. It is our intention as an instructor to prepare you as a professional who is competent in microbiological techniques. (Image: Safety Glasses by Vivien Rolfe licensed as CC BY-SA 2.0)
Upon completion of this module, you will be able to:- Obj. 1 Understand and apply biosafety as it relates to the scientific method, critical thinking and microbiology (Course Outcome # 6)
- Obj. 2 Understand and apply scientific methods and critical thinking as it relates to microbiology (C.O. #)
To achieve these objectives:
- Read the Module 1 Introduction
- Read Lab 1 Introduction and Safety. Please download and read the lecture slides about biosafety. Please download and read the “Supplemental Materials for Guidelines for Biosafety in TeachingLaboratories 2.0” by the American Society for Microbiology.
- Please complete the practice quiz about biosafety.
Note the check boxes to the right that help you track your progress: some are automatic, and some are manual.
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
Dear students, please download and read the lecture slides about biosafety.
-
Instructions
This assignment addresses course outcome(s) #6 (In a virtual setting) or (Face-to-Face setting) Perform essential microbiological techniques in a simulated environment with confidence, utilizing digital tools to analyze and interpret virtual and hands-on experimental data, and apply their virtual and/or hands-on laboratory skills to clinical and public health contexts in nursing and allied health. and module learning objective(s)
Obj. 1 Understand and apply biosafety as it relates to the scientific method, critical thinking and microbiology (Course Outcome # 6)Download and read lecture slides biosafety & ASM biosafety guidelines
Motivation:
Dear students, to the surprise and dismay of the American Society for Microbiology (ASM) biosafety guidelines for undergraduate microbiology labs were found insufficient in spite of earlier high-quality efforts. Thus, ASM developed improved and updated guidelines in 2019. As you are taking an undergraduate microbiology lab, you absolutely need to be informed about the updated guidelines.
Additionally, the lecture slide gives you information about laboratory rules, biosafety classification, good laboratory practices and some legal frameworks.
This assignment ensures in a structure manner that you get this critical information.
Instruction:
Dear student, please download the document “Supplemental Materials for Guidelines for Biosafety in Teaching Laboratories 2.0: A Revised and Updated Manual for 2019” attached as the file jmbe-20-57-s001.pdf
Dear student, please download the attached document Microbiology safety instructions v06
Please type into the text field "DONE" to confirm that you downloaded and read the material
Please do the assignment during dates of availability.
Dates of Availability: XX-XX-20XX to XX-XX-20XX
No email submissions or paper hard copy submissions will be accepted.
Contribution towards the final grade: YES
-
Dear students, please download and read Supplemental Materials for Guidelines for Biosafety in Teaching Laboratories 2.0
-
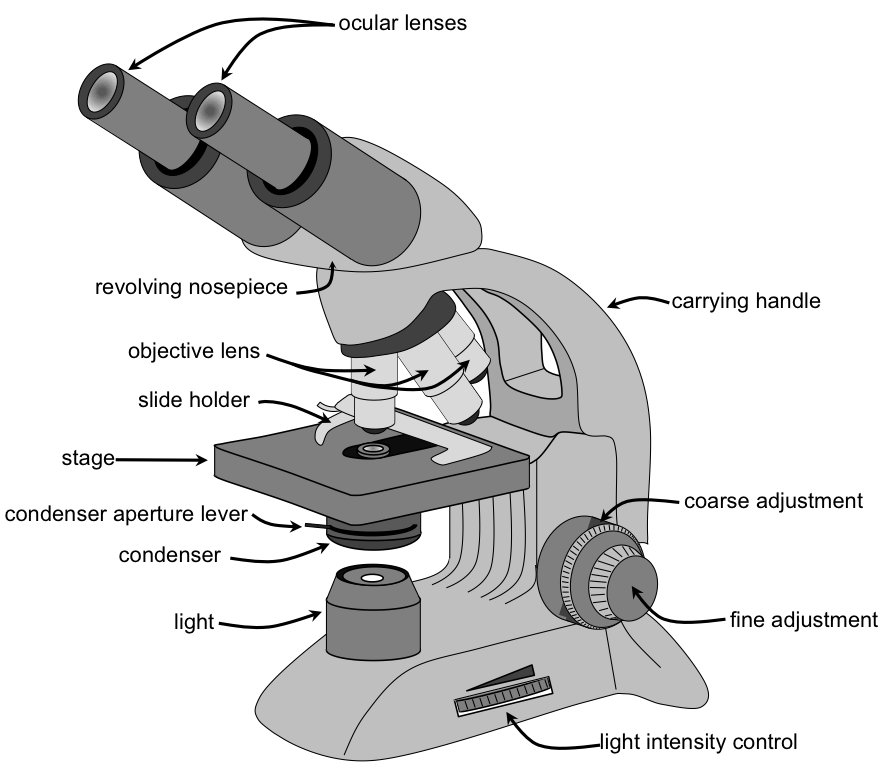 Microscopy is the scientific technique to observe objects that are too small to be seen by the naked eye. Microscopes are the tools that magnify (the process of enlarging the appearance of an object) and resolve (distinguishing two points as being separate) the minute structures such as cells, bacteria, protozoa, and their contents.
Microscopy is the scientific technique to observe objects that are too small to be seen by the naked eye. Microscopes are the tools that magnify (the process of enlarging the appearance of an object) and resolve (distinguishing two points as being separate) the minute structures such as cells, bacteria, protozoa, and their contents.An object must measure about 100 micrometers (µm) to be visible to an average human eye without a microscope. Typical animal cells measure roughly 10 µm across but are still microscopic (require a microscope to be visible). Protists, while diverse in size, generally range from 5 micrometers to 2-3 millimeters in length, Bacterial cells are typically about 1 µm, and viruses can be 10 times smaller than bacteria.
In this course, you will learn the skill of using light microscopy to identify microorganisms (too minute to see by the naked eye but visible through microscopy). You may be required to prepare wet mount slides using an environmental sample such as cultured pond water to identify eukaryotic microorganisms. Your institution may present you with prepared dry-mount slides to study the pathogenic (disease-causing) microorganisms in humans.
Upon completion of this module, you will be able to:- Identify the parts of a compound light microscope and describe their functions. (Course Objective# 1)
- Demonstrate correct use of the microscope, including focusing, adjusting lighting, and using oil immersion. (CLO#1)
- Prepare and observe wet mounts and stained slides of eukaryotic cells (e.g., protozoa, fungi, human cells).(CO#2,3)
- Differentiate between prokaryotic and eukaryotic cells based on structural features visible under the microscope.(CO#1, 4)
- Recognize common eukaryotic microorganisms, such as Candida albicans (fungus) or Amoeba and Paramecium (protozoa). (CO#4)
- Apply proper lab safety and cleaning protocols when handling biological samples and microscopes. (CO#1,3,4,5)
- Relate microscopic observations to clinical relevance in infection, immunity, and patient care. (CO#1,6)
To achieve these objectives: [Edit these items to match your resources and activities.]
- Read the Module 2 Introduction
- Read Chapter # in Textbook Title, Article Title, etc. [Include all reading assignments here that are outside of Moodle. Be as concise as possible. More information can be included in the third-party section below, if necessary.]
- Complete the [specific activities in the module. Include all in the order you want them completed. If an activity supports the development of foundational and/or industry-specific digital skills, note the skill to which it aligns]
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-

Microorganisms are found in nearly every environment on Earth, including air, water, soil, and on living organisms. In this lab, students will investigate the ubiquity of microorganisms by collecting and analyzing samples from common surfaces and environments. Through observation and comparison of microbial growth, students will gain a better understanding of how widespread microorganisms are and the factors that influence their presence and distribution.
This image shows green-blue mold colonies of Penicillium growing on culture media. Penicillium is a genus of fungi that reproduces by forming spores and can be easily distinguished from bacterial colonies by its filamentous, fuzzy appearance. U.S. Press Office. (Year). Colonies of Penicillium fungi species on agar plates [Photograph]. EurekAlert! https://www.eurekalert.org/multimedia/719840
(Image: Penicillium notatum.jpg, Wikimedia Commons, Creative Commons Attribution-Share Alike 3.0)
Upon completion of this module, you will be able to:- Explain the concept of microbial ubiquity and describe why microorganisms are found in nearly all environments.
- Demonstrate proper aseptic technique while collecting environmental samples.
- Observe and document microbial growth from different environmental sources.
- Compare microbial diversity from various surfaces or locations based on colony morphology and growth patterns.
- Interpret experimental results to explain factors that influence microbial distribution.
- Apply laboratory safety protocols when handling microbial cultures and materials.
- Communicate experimental findings clearly through written observations and analysis.
To achieve these objectives:
- Read the Module 3 Introduction
- Read Lab 3 The Ubiquity of Microorganisms: Exploring Microbial Life in Everyday Environments – Medical Microbiology for Nursing and Allied Health Lab Manual
- Complete the lab activities, quiz, and lab report
**This module could contain a midpoint check for the final project. This may include a proposal, a building block to the project, or a portion of the project.
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
 In this module you will take your midterm exam/exam # [choose one] OR submit your midterm assessment for this course.
Read the instructions carefully and take note of any special submission guidelines.
In this module you will take your midterm exam/exam # [choose one] OR submit your midterm assessment for this course.
Read the instructions carefully and take note of any special submission guidelines. NOTE: Include this module for each exam or major assessment, edited as needed. Include attribution of your image (Image: Title, Author, Source, License).
Upon completion of this module, you will have:
- Read and viewed the [midterm assessment name] instructions
- Scheduled your exam with the proctoring service [if applicable, delete if not needed]
- Prepared for and submitted your midterm assessment [revise as needed]
To achieve these objectives:
- Read and view the contents of "Exam Information and Instructions" [if applicable, delete if not needed]
- Review the [midterm assessment] guidelines in your syllabus to make sure you are ready. Click on [Title of Assessment below] and follow the instructions.
- Log in to the proctoring service and take your exam. [if applicable, delete if not needed]
-

The real worlds applications of the effect of our own various microbiomes (gut, oral, skin, vaginal, etc) are vast. Our microbiomes are affected by everything we do and eat, including diet, food additives, environmental pollutants from water, the soap we use, how much exercise we do and so on. We are not yet sure how all these changes actually manifest themselves in diseases later on, but the connections are becoming clearer. One trend is becoming clear is that the larger the diversity of any microbiome, the better and healthier the community. (Image: 25660, CDC/ Utah Department of Environmental Quality, CDC Public Health Image Library (PHIL), Public Domain).
Upon completion of this module, you will be able to:- Describe the normal microbiota and the purposes it serves in the environment and human populations.
- Describe normal and transient flora and explain proper hand-washing techniques.
- Explain different interactions between microorganisms within microbial ecological systems.
- Explain understanding of microbial life and ecology through the exploration of water samples.
- Describe how microbial processes impact microorganism distribution throughout a Winogradsky column.
- Demonstrate knowledge of complexity of microbial life within a hay infusion by discussing processes such as decomposition and growth.
To achieve these objectives:
- Read the Module 4 Introduction
- Read Chapter 4 in Medical Microbiology for Nursing and Allied Health Lab Manual.
- Read Prokaryote Slides
- Complete the Module 4 Discussion Forum
- Complete the Prokaryotic Quiz
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
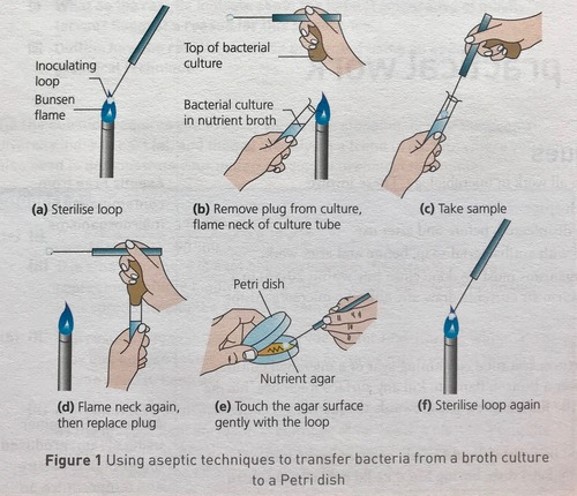
To study bacteria and other microorganisms, it is necessary to grow them in controlled conditions in the laboratory. Growth media contain a variety of nutrients necessary to sustain the growth of microorganisms. There are two commonly used physical forms of growth media: liquid media and solid growth media. A liquid medium is called a broth. Solid growth media usually contains agar, which is a mixture of polysaccharides derived from red algae. It is used as a solidification agent because it (1) is not broken down by bacteria, (2) contains no nutrients that can be used by bacteria and (3) melts at high temperatures, and yet is solid at temperatures used for most bacterial growth. Solid growth media is used in the following forms: agar plates, agar slants and agar deeps. To make agar deeps or agar slants, melted agar is poured into a test tube and then allowed to solidify vertically (agar deep), or at a slant (agar slant). Agar plates are made by pouring melted agar into a petri dish.
Upon completion of this module, you will be able to:- Demonstrate proper aseptic technique during media preparation, culture handling, and inoculation procedures (Course Outcomes #3, 5 & 6)
- Prepare common microbiological culture media following established laboratory protocols. (C.O. #3, 5 & 6)
- Explain the purpose and composition of different types of culture media and calculate the required media for preparation (general-purpose, selective, differential, and enriched media).(C.O. #3 & 5)
- Inoculate, incubate, and interpret results of bacterial cultures using aseptic techniques in face-to-face or virtual laboratory settings. (C.O. #3, 5, & 6)
- Enumerate bacterial growth using the standard plate count method and interpret results. (C.O. #3 & 6)
- Utilize digital tools or virtual laboratory platforms to simulate aseptic techniques and analyze experimental outcomes. (C.O. #6)
- Apply aseptic technique and media preparation skills to clinical and public health scenarios relevant to nursing and allied health. (C.O. #5 & 6)
- Interpret experimental results related to media preparation, contamination control, and microbial growth. (C.O. #3, 5, & 6)
To achieve these objectives:
[Welcome to this module! To be successful, please complete the activities in the sequence listed below. Each activity is designed to build your microbiology knowledge while also strengthening essential digital, scientific, and communication skills needed for today’s workforce.]
- Read the Module 5 Introduction: This provides the purpose of the module, key themes, and the learning outcomes you are expected to achieve by the end of this lab.
- Read Lab Activity Lab 5 Aseptic Techniques and Media Preparation Lisa Mims-Devezin – Medical Microbiology for Nursing and Allied Health Lab Manual
- Complete the [following activities in order. ** Please click on the fillable checklist for this laboratory Module and as you complete each activity, check off each box as you go.]
To successfully complete this laboratory module, follow each step in the order listed below. Check off each item as you complete it. Follow the steps in order. These activities are designed to help you develop safe microbiological practices, laboratory confidence, and skills applicable to clinical and public health settings.
1. Review the Laboratory Module Overview
☐ Read the Lab Module Overview page
☐ Review the importance of aseptic technique in microbiology
☐ Understand how contamination impacts patient safety, diagnostics, and research
☐ Review expectations for face-to-face or virtual lab participation
2. Review Laboratory Safety and Aseptic Technique Guidelines
☐ Review laboratory safety rules and PPE requirements
☐ Review principles of aseptic technique
☐ Review proper use of flame, biosafety cabinets, and sterile work areas
3. Watch the Aseptic Technique Demonstration Videos
☐ Introduction to Aseptic Technique
☐ Proper Handling of Cultures and Media
☐ Sterilization of Tools and Work Areas
☐ Preventing Contamination During Transfers
☐ Foundational Digital Skill: Digital Content Navigation
4. Complete the Media Preparation Reading
☐ Review types of culture media
☐ Review media components and their functions
☐ Review sterilization methods
5. Perform the Media Preparation Activity
☐ Prepare culture media following laboratory protocol (face-to-face or virtual simulation)
☐ Label media correctly (name, date, medium type)
☐ Sterilize media using approved methods
☐ Industry-Specific Digital Skill (Virtual): Laboratory Simulation Tools
☐ Foundational Digital Skill: Procedural Accuracy in Digital Environments
6. Demonstrate Aseptic Transfer Techniques
☐ Inoculate prepared media with provided cultures
☐ Transfer microorganisms between media without contamination
☐ Properly incubate cultures according to protocol
7. Perform the Standard Plate Count Activity
☐ Prepare serial dilutions
☐ Plate diluted samples using aseptic technique
☐ Count colonies and calculate CFU/mL
☐ Foundational Digital Skill: Quantitative Reasoning Using Digital Tools
8. Record and Analyze Experimental Results
☐ Record observations and measurements
☐ Identify possible sources of contamination
☐ Interpret growth patterns and plate count results
☐ Foundational Digital Skill: Digital Data Entry and Analysis
9. Complete the Laboratory Assessment
☐ Complete the lab quiz covering aseptic technique, media preparation, and plate counts
☐ Foundational Digital Skill: Digital Assessment Tools
10. Submit the Laboratory Report or Worksheet
☐ Upload completed lab worksheet or report
☐ Include calculations, interpretations, and screenshots if applicable
11. Review Instructor Feedback
☐ Review instructor feedback using the LMS grading tool
☐ Foundational Digital Skill: Using Digital Feedback Tools
12. Complete the Laboratory Reflection
☐ Respond to the reflection prompt on aseptic technique and patient safety
☐ Industry-Specific Digital Skill: Reflective Practice in Healthcare Education
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
This file contains the PowerPoint Slides for Module 5 Lab Activity on Aseptic Technique and Media Preparation. I am also including this file as a PDF version. You can locate the PDF version just below this file.
-
This file contains the PDF version of the Laboratory Module 5: Aseptic Technique and Media Preparation PowerPoint slides. This PDF file provides access to the slides if Microsoft PowerPoint is not readily available. Thank you.
- Demonstrate proper aseptic technique during media preparation, culture handling, and inoculation procedures (Course Outcomes #3, 5 & 6)
-
Capsule staining is a specialized microbiological technique used to visualize the gelatinous outer layer, or capsule, that surrounds certain bacterial cells. Unlike many staining procedures that color the cell itself, capsule staining relies on contrasting the background and the cell to reveal the presence of the capsule as a clear, halo-like zone. This structure, typically composed of polysaccharides or polypeptides, plays an important role in bacterial survival by aiding in adhesion, protecting cells from desiccation, and helping pathogenic bacteria evade host immune defenses such as phagocytosis. In this module, students will explore the structure and function of bacterial capsules, learn the principles behind capsule staining, and develop the skills needed to correctly perform the procedure and interpret stained specimens using the microscope.
Slide prepared and image captured by Professor Erica A. Burns, Fletcher Technical Community College.
Course Learning Objectives1. Distinguish between Bright-field and Dark-field Microscopy
2. Understand Staining Techniques
3. Prepare Culture Media and Cultivate Bacteria
4. Identify Unknown Bacteria Using Physiological Tests
5. Demonstrate Basic Culture Techniques in Healthcare Settings
6. Perform Essential Microbiological Techniques in a Virtual Setting
Upon completion of this module, you will be able to:
- Describe the structure and functions of the capsule and its benefit to pathogenic bacteria (CO#2)
- Explain the basic principles for the capsule stain (CO#2)
- List clinically important pathogenic organisms that have a capsule (CO#5)
- Demonstrate how to preform a capsule stain (CO#2,3)
- Interpret the results of a capsule stained slide using a microscope (CO#4)
- Draw, label and describe the location of the capsule (CO#2)
To achieve these objectives:- Read the Module 6 Introduction
- Read Lab 6 Capsule Staining – Medical Microbiology for Nursing and Allied Health Lab Manual
- Review Capsule Staining PowerPoint
- Complete the Module 6 Discussion Forum
- Complete the Lab 6 Capsule Stain Post Lab Questions
**Please note: the final project should be submitted in this module or the next. Consider this when assigning activities and for grading time.
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
Note to instructors:
These are the questions in the ebook.
None of the setting have been set so make sure your set them to your liking.
Objectives that link to each question are in the question feedback section.
-
Endospores are dormant forms of living bacteria and should not be confused with reproductive spores produced by fungi. These structures are produced by a few genera of Gram-positive bacteria, almost all bacilli, in response to adverse environmental conditions. Two common bacteria that produce endospores are Bacillus or Clostridum. Both live primarily in soil and as symbionts of plants and animals, and produce endospores to survive in an environment that change rapidly and often.
The process of endosporulation (the formation of endospores) involves several stages. After the bacterial cell replicates its DNA, layers of peptidoglycan and protein are produced to surround the genetic material. Once fully formed, the endospore is released from the cell and may sit dormant for days, weeks, or years. When more favorable environmental conditions prevail, endospores germinate and return to active duty as vegetative cells..Upon completion of this module, you will be able to:
- Define the term endospore and explain its biological significance in bacterial survival, dormancy, and resistance. (C.O. # 4, 5, & 6)
- Differentiate between vegetative cells and endospores based on morphology and staining characteristics. (C.O. #2, 4, 5, & 6)
- Identify bacterial genera commonly associated with endospore formation (e.g., Bacillus, Clostridium). (C.O. #4, 5, & 6)
- Describe the principles and steps of the Schaeffer-Fulton endospore staining technique, including the function of:
• Malachite green (primary stain)
• Heat (mordant to drive stain into endospore)
• Water (decolorizer)
• Safranin (counterstain for vegetative cells) (C.O. #2, 5, & 6) - Demonstrate proper aseptic technique in preparing a bacterial smear for staining. (C.O. #5 & 6)
- Perform the complete endospore staining protocol with attention to:
• Heat fixing the smear appropriately
• Using steam to enhance dye uptake into endospores
• Rinsing and counterstaining effectively (C.O. #2, 5, & 6) - Observe and interpret stained specimens under oil immersion (1000× total magnification), distinguishing between:
• Green-stained endospores
• Red/pink vegetative cells.(C.O. #1, 2, 5, & 6) - Explain the clinical or environmental importance of endospore-forming bacteria in contexts such as:
• Disease transmission (e.g., Clostridioides difficile, Bacillus anthracis)
• Food and equipment sterilization
• Infection control.(C.O. #4, 5, & 6) - Record and analyze staining results in laboratory notebooks using appropriate scientific terminology and diagrammatic representation. (C.O. #5 & 6)
- Use a digital tool to create a chart of the most commonly isolated endospore-former(s) in your city and state. (C.O. #4 & 6)
- Using a digital tool, create a concept map for a selected endospore-forming microorganism. (C.O. #4 & 6)
To achieve these objectives:
[Welcome to this module! To be successful, please complete the activities in the sequence listed below. Each activity is designed to build your microbiology knowledge while also strengthening essential digital, scientific, and communication skills needed for today’s workforce.]
- Read the Module 7 Introduction: This provides the purpose of the module, key themes, and the learning outcomes you are expected to achieve by the end of this lab.
- Read Lab 7 Endospore Staining – Medical Microbiology for Nursing and Allied Health Lab Manual
- Complete the [following activities in order. ** Please click on the fillable checklist for this laboratory Module and as you complete each activity, check off each box as you go.]
1. Module Orientation
☐ Read the Endospore Stain Laboratory Module Overview.
2. Laboratory Safety & Aseptic Technique
☐ Review laboratory safety rules and PPE requirements.
☐ Review aseptic technique principles for smear preparation.
☐ Review safe use of heat sources and staining reagents.
3. Watch the Endospore Stain Demonstration Videos
☐ Introduction to endospores and their significance.
☐ Overview of the Schaeffer–Fulton endospore stain.
☐ Preparing and heat-fixing a bacterial smear.
☐ Performing the endospore staining procedure.
☐ Microscopy and interpretation of results.
☐ Foundational Digital Skill: Digital Content Navigation.
4. Complete the Required Reading: Endospore Stain
☐ Differences between vegetative cells and endospores.
☐ Endospore-forming genera (e.g., Bacillus, Clostridium).
☐ Principles of the Schaeffer–Fulton method: malachite green, heat, water, safranin.
5. Prepare the Bacterial Smear
☐ Prepare a bacterial smear using proper aseptic technique.
☐ Air-dry and heat-fix the smear according to protocol.
☐ Industry-Specific Digital Skill (Virtual): Laboratory Simulation Tools.
☐ Foundational Digital Skill: Procedural Accuracy in Digital Environments.
6. Perform the Endospore Staining Procedure
☐ Apply malachite green and steam to drive stain into endospores.
☐ Rinse with water to decolorize vegetative cells.
☐ Apply safranin to counterstain vegetative cells.
7. Microscopy & Interpretation
☐ Observe stained specimens using oil immersion (1000×).
☐ Identify green endospores.
☐ Identify red/pink vegetative cells.
8. Record & Analyze Laboratory Results
☐ Record staining results using the lab worksheet or digital form.
☐ Include labeled diagrams or drawings.
☐ Interpret the significance of endospore presence.
9. Complete the Laboratory Assessment
☐ Complete the endospore stain lab quiz.
☐ Foundational Digital Skill: Digital Assessment Tools
10. Digital Data Activity Assignment
☐ Use a digital tool to create a chart of the most commonly isolated endospore-forming bacteria in your city and state.
☐ Foundational Digital Skill: Digital Assessment Tools
11. Digital Concept Mapping Assignment
☐ Select one endospore-forming microorganism.
☐ Create a digital concept map including morphology, endospore formation, and clinical or environmental significance.
☐ Foundational Digital Skill: Digital Assessment Tools
12. Submit Your Laboratory Assignments
☐ Upload completed lab worksheet or report.
☐ Upload digital chart.
☐ Upload digital concept map.
☐ Confirm all files are labeled with your name.
13. Review Instructor Feedback
☐ Review instructor feedback using the LMS grading tool.
☐ Foundational Digital Skill: Digital Assessment Tools
14. Complete the Laboratory Reflection
☐ Respond to the reflection prompt on endospore-forming bacteria and public health.
☐ Industry-Specific Digital Skill: Reflective Practice in Healthcare Education
Module Pressbooks Resources and ActivitiesYou will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
This file contains the PowerPoint Slides for Module 7 Lab on the Endospore Stain. I am also including this file as a PDF version. You can locate the PDF version just below this file.
-
This file contains the PDF version of the Laboratory Module 7: The Endospore Stain PowerPoint slides. This PDF file provides access to the slides if Microsoft PowerPoint is not readily available. Thank you.
-
This YouTube video demonstrates how to perform the Endospore Stain. Please review this video as an ancillary tool for this laboratory activity.
- Define the term endospore and explain its biological significance in bacterial survival, dormancy, and resistance. (C.O. # 4, 5, & 6)
-

In this module you will take your final exam/submit your final assessment [choose one] for this course. Read the instructions carefully and take note of any special submission guidelines.
To replace image, double click the image placeholder. Select 300–400 pixels wide, and update the description of image. Include attribution of your image (Image: Title, Author, Source, License).Upon completion of this module, you will have:
- Read and viewed the final assessment instructions
- Scheduled your exam with the proctoring service [if applicable]
- Prepared and submitted your final assessment
To achieve these objectives:
- Read and view the contents of the module book "Exam Information and Instructions" [if exam only]
- Review the final assessment guidelines in your syllabus to make
sure you are ready. Click on [Title of Assessment below] and follow the instructions.
- Log in to the proctoring service and take your exam. [if exam only]
-
Acid-fast staining is a specialized differential staining technique used to identify bacteria with unique cell walls that contain high concentrations of waxy lipids, particularly mycolic acids. These lipids make the cells resistant to conventional staining methods and allow them to retain the primary stain even after treatment with strong decolorizing agents. This characteristic is especially important for the identification of clinically significant organisms, such as species of Mycobacterium. In this module, students will examine the structural basis of acid-fastness, learn the principles behind the acid-fast staining procedure, and practice performing and interpreting an acid-fast stain to distinguish acid-fast bacteria from non–acid-fast organisms using the microscope.
Slide made by Fletcher Technical Community College student Nicola Ladner. Image captured by Professor Erica A. Burns
Course Learning Objectives
1. Distinguish between Bright-field and Dark-field Microscopy
2. Understand Staining Techniques
3. Prepare Culture Media and Cultivate Bacteria
4. Identify Unknown Bacteria Using Physiological Tests
5. Demonstrate Basic Culture Techniques in Healthcare Settings
6. Perform Essential Microbiological Techniques in a Virtual Setting
Upon completion of this module, you will be able to:- Explain the purpose of the acid-fast stain. (CO#2)
- Identify the cell wall substance unique to acid-fast bacteria that allows them to retain the carbolfuchsin stain. (CO#2)
- Identify the decolorizer used in the acid-fast staining procedures. (CO#2)
- List groups of pathogenic organisms that stain acid-fast. (CO#5)
- Perform a Ziehl–Neelsen acid-fast stain. (CO#2,3,4)
- Distinguish acid-fast (positive) from non-acid-fast (negative) bacteria on a slide using a microscope. (CO#4,6)
To achieve these objectives:
- Read the Module # Introduction
- Read and view the materials in the Module #8, Acid-Fast Staining PowerPoint, and Pressbooks book chapter
- Read Lab 8 in Medical Microbiology for Nursing and Allied Health Lab Manual
- Complete the Lab 8 Acid-Fast Post Lab Questions Quiz
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
 Gram staining is a fundamental laboratory technique used in microbiology to classify bacteria based on the structural differences in their cell walls. Developed by Hans Christian Gram in 1884, this differential staining method divides bacteria into two main categories: Gram-positive and Gram-negative.
Gram staining is a fundamental laboratory technique used in microbiology to classify bacteria based on the structural differences in their cell walls. Developed by Hans Christian Gram in 1884, this differential staining method divides bacteria into two main categories: Gram-positive and Gram-negative.The classification is determined by the bacteria's ability to retain the primary stain, crystal violet, following a decolorization step. Gram-positive bacteria appear purple due to their thick peptidoglycan layer, while Gram-negative bacteria appear pink or red due to a thinner peptidoglycan layer and an outer membrane. Gram staining is a critical first step in identifying bacterial pathogens and plays a key role in guiding initial antibiotic therapy and infection control practices in clinical settings.
Course Learning Objectives
1. Distinguish between Bright-field and Dark-field Microscopy
2. Understand Staining Techniques
3. Prepare Culture Media and Cultivate Bacteria
4. Identify Unknown Bacteria Using Physiological Tests
5. Demonstrate Basic Culture Techniques in Healthcare Settings
6. Perform Essential Microbiological Techniques in a Virtual Setting
Upon completion of this module, you will be able to:
- Explain the basic principles behind the Gram staining procedure, including the role of crystal violet, iodine, ethanol, and safranin. (CO#2)
- List and describe each step of the Gram staining process, including application of crystal violet, iodine treatment, decolorization, and counterstaining with safranin. (CO#2,3)
- Explain the differences in cell wall structure between Gram-positive and Gram-negative bacteria and how these differences influence the Gram staining results. (CO#2 )
- Identify and interpret the color and morphology of bacteria after Gram staining, recognizing Gram-positive organisms as purple and Gram-negative organisms as pink. (CO#4)
- Demonstrate how Gram staining can be used as a preliminary method to identify bacterial species and guide further diagnostic testing. (CO#4,5)
- Discuss common errors or factors that can affect the accuracy of Gram staining results, such as over-decolorization or under-staining. (CO#3,5)
- Recognize and follow proper laboratory safety and handling procedures when performing Gram staining. (CO#3,5,6)
To achieve these objectives:
- Read the Module #9 Introduction
- Read and view the materials in the Module #9, Gram Staining PowerPoint, and Pressbooks book chapter
- Read Lab 9 in Medical Microbiology for Nursing and Allied Health Lab Manual
- Complete the Module 9 Assignment
- Complete the Module 9 Discussion Forum
-
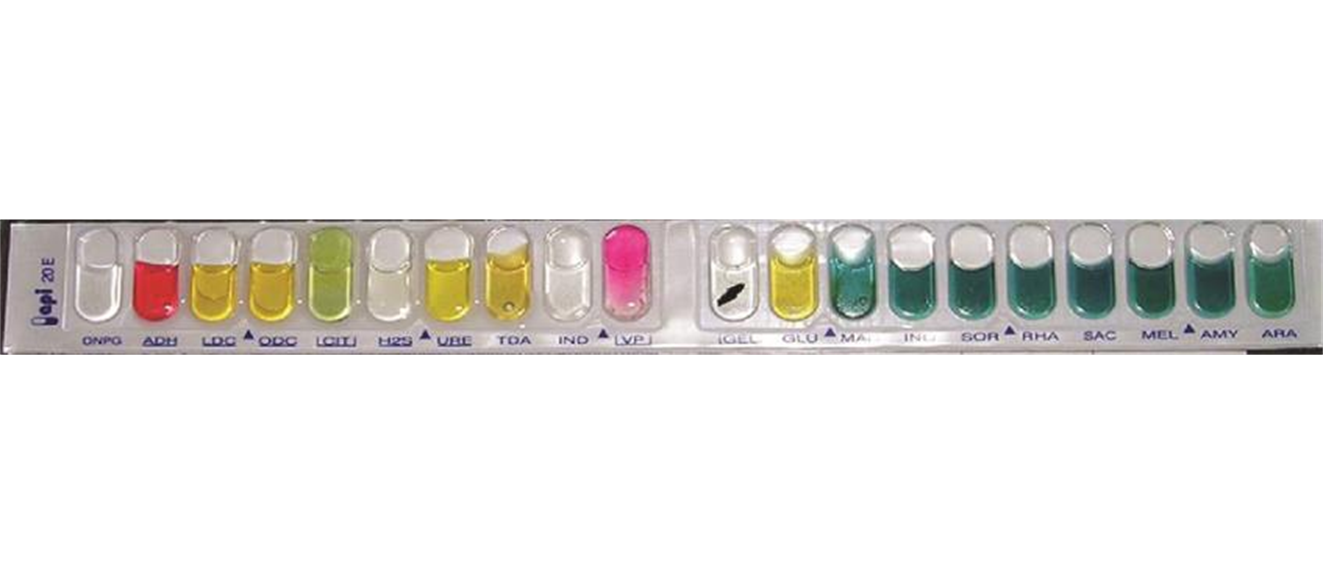
A multiplexed biochemical test such as API 20 E is shown. The API 20NE test strip is used to identify specific strains of gram-negative bacteria outside the Enterobacteriaceae. Here is an API 20NE test strip result for Photobacterium damselae ssp. piscicida. (Source of Image: Figure 8.18, OpenStax Microbiology, CC BY License)
Biochemical testing is based on the fact that microbes have microbial metabolism involving microbial biochemistry. Different microbes have different metabolic capabilities. Biochemical testing can therefore be used to characterize an unknown microbe and figure out the identity of the microbes. Characterizing and identifying an unknown microbe based on biochemical testing is not only an intellectual exercise for a microbiologist. Characterizing and identifying an unknown microbe based on biochemical testing is in healthcare a critical task to determine the identity of the pathogen causing illness in the patient and finding the appropriate therapeutic intervention. Biochemical tests for fermentation, gelatin hydrolysis, starch hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophane hydrolysis are some examples.Upon completion of this module, you will be able to:
- Obj. 1 Be
able to perform the following biochemical tests and understand how they work:
carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein
hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan
hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various
physiological tests.)
- Obj. 2 Be
able to relate the results of the tests performed to the different types of
metabolic activities carried out by individual bacterial species. (C.O. # 4 Identify
unknown bacteria using various physiological tests.)
To achieve these objectives:
- Read the Module #10 Introduction
- Read
and view the materials in the Module “LAB
10: BIOCHEMICAL TESTING” in the Pressbooks book. It shows commonly used
biochemical tests for fermentation, gelatin hydrolysis, starch
hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase
activity, tryptophane hydrolysis.
- Read Chapter #10 in Medical Microbiology for Nursing and Allied Health Lab Manual.
- Complete the following specific activities in the module:
- Download and read the lecture slides about fermentation (file fermentation v08)
- Take the quiz about fermentation.
- Download and read the lecture slides about hydrolytic and degradative enzymes (file Hydrolytic _degradative enzymes v09b)
- Take the quiz about hydrolytic and degradative enzymes (Hydrolysis)
- Download and read the lecture slides about urea – hydrolytic reactions (file hydrolytic reactions - urea v02b)
- Take the quiz about urease
- Download and read the lecture slides about catalase and oxidase test (file biooxidation & respiration v09b)
- Take the quiz about catalase and oxidase test
- The instructor will determine which lab test you will actually conducting in the lab from the book Module “LAB 10: BIOCHEMICAL TESTING”
- Assignment: Upload the lab report of the actual assigned lab test that your instructor assigned you to perform.
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
Dear students,
Please download and read the lecture slides about fermentation (file fermentation v08).
-
Dear students,
Please download and read the lecture slides about urea – hydrolytic reactions (file hydrolytic reactions - urea v02b)
-
Dear students,
Please download and read the lecture slides about hydrolytic and degradative enzymes (file Hydrolytic _degradative enzymes v09b).
-
Dear students,
Please download and read the lecture slides about catalase and oxidase test (file biooxidation & respiration v09b)
-
Dear students,
The quiz is aligned with
- Obj. 1 Be able to perform the following biochemical tests and understand how they work: carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various physiological tests.)
- Obj. 2 Be able to relate the results of the tests performed to the different types of metabolic activities carried out by individual bacterial species. (C.O. # 4 Identify unknown bacteria using various physiological tests.)
Please take quiz/assignment during dates of availability:
Dates of availability: xx/xx/202x to xx/xx/202x
Attempts: unlimited within dates of availability.
Grade/score: Highest score of attempt counts.
Length of time per attempt: You have xxx minutes for each attempt.
Uninterrupted attempts required.
Number of questions: There are 14 questions.
Contribution towards final grade: yes/no
The quiz has to be taken in the learning management system, in this case Canvas or Moodle.
Email, paper, or any other type of submissions will not be accepted.
Sincerely,
-
Dear students,
The quiz is aligned with
- Obj. 1 Be able to perform the following biochemical tests and understand how they work: carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various physiological tests.)
- Obj. 2 Be able to relate the results of the tests performed to the different types of metabolic activities carried out by individual bacterial species. (C.O. # 4 Identify unknown bacteria using various physiological tests.)
Please take quiz/assignment during dates of availability:
Dates of availability: xx/xx/202x to xx/xx/202x
Attempts: unlimited within dates of availability.
Grade/score: Highest score of attempt counts.
Length of time per attempt: You have xxx minutes for each attempt.
Uninterrupted attempts required.
Number of questions: There are 18 questions.
Contribution towards final grade: yes/no
The quiz has to be taken in the learning management system, in this case canvas or Moodle.
Email, paper, or any other type of submissions will not be accepted.
Sincerely,
Your instructor - Obj. 1 Be able to perform the following biochemical tests and understand how they work: carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various physiological tests.)
-
Dear students,
The quiz is aligned with
- Obj. 1 Be able to perform the following biochemical tests and understand how they work: carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various physiological tests.)
- Obj. 2 Be able to relate the results of the tests performed to the different types of metabolic activities carried out by individual bacterial species. (C.O. # 4 Identify unknown bacteria using various physiological tests.)
Please take quiz/assignment during dates of availability:
Dates of availability: xx/xx/202x to xx/xx/202x
Attempts: unlimited within dates of availability.
Grade/score: Highest score of attempt counts.
Length of time per attempt: You have xxx minutes for each attempt.
Uninterrupted attempts required.
Number of questions: There are 16 questions.
Contribution towards final grade: yes/no
The quiz has to be taken in the learning management system, in this case canvas or Moodle.
Email, paper, or any other type of submissions will not be accepted.
Sincerely,
Your instructor - Obj. 1 Be able to perform the following biochemical tests and understand how they work: carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various physiological tests.)
-
Dear students,
the quiz is aligned with
- Obj. 1 Be able to perform the following biochemical tests and understand how they work: carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various physiological tests.)
- Obj. 2 Be able to relate the results of the tests performed to the different types of metabolic activities carried out by individual bacterial species. (C.O. # 4 Identify unknown bacteria using various physiological tests.)
Please take quiz/assignment during dates of availability:
Dates of availability: xx/xx/202x to xx/xx/202x
Attempts: unlimited within dates of availability.
Grade/score: Highest score of attempt counts.
Length of time per attempt: You have xxx minutes for each attempt.
Uninterrupted attempts required.
Number of questions: There are 8 questions.
Contribution towards final grade: yes/no
The quiz has to be taken in the learning management system, in this case Canvas or Moodle.
Email, paper, or any other type of submissions will not be accepted.
Sincerely,
Your instructor - Obj. 1 Be able to perform the following biochemical tests and understand how they work: carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various physiological tests.)
-
Opened: Tuesday, December 23, 2025, 12:00 AMDue: Tuesday, December 30, 2025, 12:00 AM
The assignment is aligned with:
- Obj. 1 Be able to perform the following biochemical tests and understand how they work: carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various physiological tests.)
- Obj. 2 Be able to relate the results of the tests performed to the different types of metabolic activities carried out by individual bacterial species. (C.O. # 4 Identify unknown bacteria using various physiological tests.)
Assignment: Upload the lab report of the actual assigned lab test that your instructor assigned you to perform (to be announced).
The instructor will determine which lab test you will actually conducting in the lab from the book Module “LAB 10: BIOCHEMICAL TESTING”
Upload the lab report of the actual assigned lab test that your instructor assigned you to perform.
The structure of the lab report depends on requirements of the instructor.
Motivation:
Motivation for the lab report is that students experience and demonstrate the skills 1) to apply the scientific method, 2) apply standard techniques in microbiology, 3) process and understand the underlying theoretical concepts of microbiology.
Instruction:
One format for a lab report is to report the 1) title of the experiment or test, 2) the hypothesis or research question of the experiment or test, 3) the prediction, 4) the procedure, 5) the results, and 6) the conclusion.
Example of a lab report -
1) title of the experiment or test: Catalase test
2) the hypothesis or research question of the experiment or test: It is hypothesized that the bacteria to be tested will be catalase positive OR the research question studied is whether or not the tested bacteria are calatase positive
3) the prediction: If the bacteria are catalase positive, then formation of bubbles will be observed during the test
4) the procedure: Transfer bacteria of interest onto microscopic slide using aseptic techniques. Apply enough droplets of 3% hydrogen peroxide solution, so that microbes are covered. Observe microbes in 3% hydrogen peroxide solution for 1 to 3 minutes for the formation of bubbles. Document the results. In parallel, include a positive control of a known lab strain that is positive for catalase
5) the results:
The bacteria of interest did/ did not form bubbles (circle appropriate result).
The positive control did/did not form bubbles (circle appropriate result).
Include photos if available.
6) the conclusion:
The positive control did/did not work , because the bubbles were/were not formed (circle appropriate result). Therefore, the test reagents and the test itself is/is not working properly (circle appropriate result).
The unknown/bacteria of interest are/are not catalase positive, because the bacteria did/did not form bubbles (circle appropriate result). The results do/don’t support the hypothesis (circle appropriate result).
You can upload your report as a file, or write by hand on a piece of paper and upload a photo of your handwritten report.
- Obj. 1 Be able to perform the following biochemical tests and understand how they work: carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various physiological tests.)
- Obj. 1 Be
able to perform the following biochemical tests and understand how they work:
carbohydrate fermentation, gelatin hydrolysis, starch hydrolysis, casein
hydrolysis, urea hydrolysis, citrate utilization, catalase activity, tryptophan
hydrolysis (indole test). (C.O. # 4 Identify unknown bacteria using various
physiological tests.)
-
 In your previous studies, you’ve learned the basics of bacterial growth and how microbes cause infections. This module builds on that knowledge by examining how physical factors like temperature, pH, and osmotic pressure affect bacterial growth and survival. Through hands-on experiments measuring bacterial growth over time, you’ll explore how different bacteria adapt to various environments—such as heat-loving or acid-loving microbes—and how these conditions influence infection risks. From a nursing perspective, understanding these concepts is essential because it helps you appreciate how environmental factors can affect pathogen behavior and patient outcomes. This knowledge equips you to better interpret lab results, support infection prevention strategies, and contribute to effective patient care and antibiotic stewardship in your future nursing career.
In your previous studies, you’ve learned the basics of bacterial growth and how microbes cause infections. This module builds on that knowledge by examining how physical factors like temperature, pH, and osmotic pressure affect bacterial growth and survival. Through hands-on experiments measuring bacterial growth over time, you’ll explore how different bacteria adapt to various environments—such as heat-loving or acid-loving microbes—and how these conditions influence infection risks. From a nursing perspective, understanding these concepts is essential because it helps you appreciate how environmental factors can affect pathogen behavior and patient outcomes. This knowledge equips you to better interpret lab results, support infection prevention strategies, and contribute to effective patient care and antibiotic stewardship in your future nursing career.Image: Adapted from Petersen J. & McLaughlin S., Laboratory Exercises in Microbiology: Discovering the Unseen World Through Hands-On Investigation, Queensborough Community College, Fall 2016. Licensed under CC BY-NC-SA 4.0.” (Petersen & McLaughlin, 2016)
Upon completion of this module, you will be able to:
- Explain the impact of temperature on microbial growth (Linked COs: 1)
- Describe microbial adaptations to different pH environments (Linked COs: 1)
- Analyze the effects of osmotic pressure on bacterial cells (Linked COs: 1)
- Read and understand the lab manual
- Read Chapter 7 in Microbiology for Nursing/Allied Health
- Complete the H5P questions in the lab manual
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-

As future nurses, understanding how antibiotics work and how we determine their effectiveness is essential to providing safe, evidence-based care. This module builds on your existing knowledge of antibiotics by introducing the Kirby-Bauer disk diffusion test, a standard lab method used to identify which antibiotics are most effective against specific bacterial infections. You’ll learn how to interpret lab results that classify bacteria as susceptible, intermediate, or resistant—information that directly impacts treatment decisions at the bedside. With antibiotic resistance on the rise, this knowledge is crucial for supporting proper medication use, improving patient outcomes, and contributing to responsible antibiotic stewardship in clinical practice.
Image: Adapted from “Disk Diffusion Method for Antibiotic Susceptibility Test” (Creative Biolabs, 2025).
Upon completion of this module, you will be able to:
Explain the principle and clinical relevance of the Kirby-Bauer disk diffusion method (Linked COs: 3).
Properly perform antibiotic susceptibility testing using standardized inoculation and disk placement techniques (Linked COs: 3).
Accurately measure zones of inhibition and interpret results using CLSI guidelines (Linked COs: 3).
Classify antibiotics by their mechanism of action and relate this to their effectiveness against specific bacteria (Linked COs: 2, 3).
To achieve these objectives:
- Read and understand the lab manual
- Read Chapter 8 in Microbiology for Nursing/Allied Health
- Complete the H5P questions in the lab manual
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
.png?revision=1)
Food products provide rich environments for microbial growth. In this lab, students examine microorganisms found in milk, poultry, and other foods using serial dilution and standard plate count techniques. The lab emphasizes food safety, microbial indicators, and the importance of proper food handling.
Typical growth on EMB. Left - a non-lactose fermentor. Right - a vigorous lactose fermenter such as E. coli produces a characteristic green sheen. BioLibreTexts. https://bio.libretexts.org/Courses/City_College_of_San_Francisco/Microbiology_Lab_Manual_%28Miraglia%29/01%3A_New_Page/1.12%3A_Completion_of_the_ID_ProjectUpon completion of this module, you will be able to:
- Explain how microorganisms contaminate food
- Perform serial dilutions and standard plate counts
- Interpret CFU data to estimate microbial load
- Relate microbial presence to food safety practices
To achieve these objectives:
- Read the Module 13 Introduction
- Read and view the materials in the Module #13 Pressbooks book
- Read Lab #13 in Medical Microbiology for Nursing and Allied Health Lab Manual
- Complete the discussion board, quiz, and lab report.
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
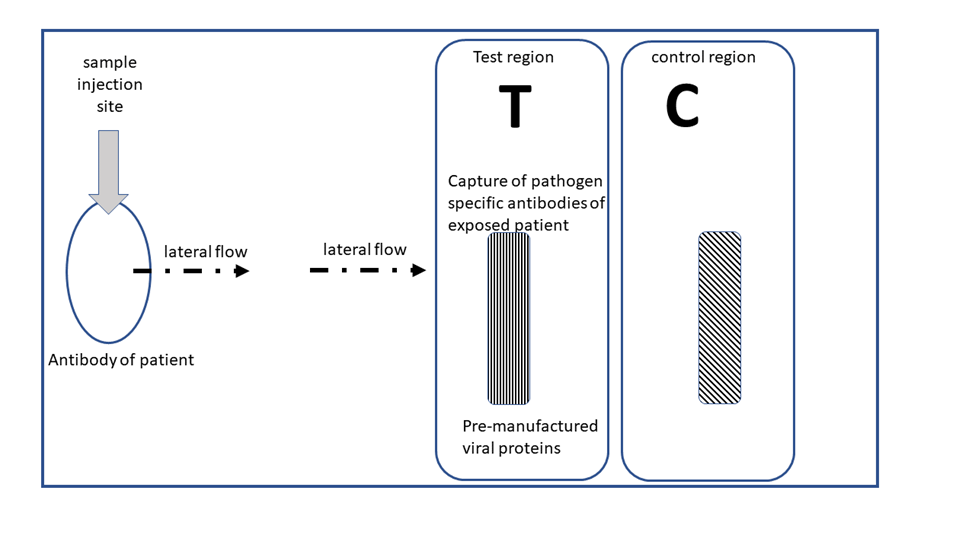
Recent advances in biotechnology have given rise to new rapid diagnostic testing methods in microbiology and human health. As a patient going to urgent care or doctor’s office you may have encountered rapid diagnostic tests for detecting the influenza virus or the virus causing COVID19. Most likely, you may use a rapid diagnostic test as an allied health professional at some time in the future. This lab module gives you some information about rapid diagnostic tests to prepare you better for your professional career. Content areas about rapid diagnostic tests include: the advantages, uses, working principles, and details of lateral flow immunochromatography. (Image: Schematic of a positive test result of a lateral flow immunochromatography test for a virus. By Illya Tietzel, 2025 Creative Commons 4.0 BY).
Upon completion of this module, you will be able to:
- Obj. 1 Remember advantages of rapid diagnostic testing (C.O. #4: Identify unknown bacteria using various
physiological tests.) & (C.O. # 6 : In a virtual setting) or (Face-to-Face
setting) Perform essential microbiological techniques in a simulated
environment with confidence, utilizing digital tools to analyze and interpret
virtual and hands-on experimental data, and apply their virtual and/or hands-on
laboratory skills to clinical and public health contexts in nursing and allied
health.)
- Obj. 2 Distinguish between different uses of rapid diagnostic tests (C.O. # 4 & C.O. # 6)
- Obj. 3 Distinguish between the different working principles of different rapid diagnostic tests (C.O. # 4 & C.O. # 6).
- Obj. 4 Identify
positive, negative, and erroneous or inclusive test result for lateral flow
immunochromatography (C.O. # 4 & C.O. # 6)
- Obj. 5 Distinguish
between the different working principles of different rapid diagnostic tests (C.O. # 4 & C.O. # 6)
To achieve these objectives:
- Read the Module #14
Introduction
- Read and view the materials
in the Module # Lab 14 Rapid
Diagnostic of the Pressbooks book
- Please download and read the lecture slides about Rapid Diagnostic tests.
- Complete the
- quiz entitled “Quiz about rapid diagnostic tests”
- Module #14 Assignment Assignment Pubmed Central search for articles about lateral flow chromatography and detection of viruses (Basic Digital Literacy: Perform Online Searches on a scientific website for biomedical literature to find accurate and relevant information: Assignment Pubmed Central search for articles about lateral flow chromatography and detection of viruses).
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
-
Dear students,
please download and read the lecture slides.
-
Motivation:
This assignment addresses course outcome(s) #6 (In a virtual setting) or (Face-to-Face setting) Perform essential microbiological techniques in a simulated environment with confidence, utilizing digital tools to analyze and interpret virtual and hands-on experimental data, and apply their virtual and/or hands-on laboratory skills to clinical and public health contexts in nursing and allied health and module learning objective(s) Obj. 5 Distinguish between the different working principles of different rapid diagnostic tests. It also addresses basic digital literacy skills such as performing online searches to find accurate and relevant information using search engines.
Allied Health professional should be able to find accurate information about biomedical topics using scientific search engines such as Pubmed or PubMed Central.
As the module was aiming to increase the knowledge base of students about rapid diagnostic tests such as lateral flow immuno-chromatography, students will use PubMed Central to find information about lateral flow immuno-chromatography and detection of viruses.
Instruction:
Open an internet browser. Type into the search engine the words:” pubmed central”.
The search result should show the website or URL for PubMed Central. It is [https://pmc.ncbi.nlm.nih.gov/].
Clicking on the link a new website should open.
Into the search bar, the students paste phrase: lateral flow immunochromatography AND detection of viruses.
The results page should show about 9,015 biomedical articles.
The students should then select one article of their choice and upload the information into the assignment. Please upload the title of the article, the names of the authors, and the information about the scientific journal or book.
For example, a fictitious student has chosen this article:
- Title: A novel diagnostic approach to detecting porcine epidemic diarrhea virus: The lateral immunochromatography assay.
- Authors: Kim YK, Lim SI, Cho IS, Cheong KM, Lee EJ, Lee SO, Kim JB, Kim JH, Jeong DS, An BH, An DJ.
- Name of Journal or book: J Virol Methods. 2015 Sep 2;225:4-8. doi: 10.1016/j.jviromet.2015.08.024.
More background information: Our original search phrase ( lateral flow immunochromatography AND detection of viruses) had the word and in capital letters as AND. This is on purpose. The PubMed Central search engine considers AND as a command. The command means to combine the search terms before and after AND. In this case we combined lateral flow immunochromatography with the detection of viruses.
Evaluation rubric:
Expectations met (100 points) – a scientific article was uploaded with title of the article, the names of the authors, and the name of the journal or book
Expectations partially met (70 points) – 2 out of 3 elements (title, authors, name of journal or book) were uploaded.
Expectations mostly incomplete (25 points) - 1 out of 3 elements (title, authors, name of journal or book) were uploaded.
Expectations mostly incomplete (0 points) - 0 out of 3 elements (title, authors, name of journal or book) were uploaded.
Students can upload files as text or word processing files in the format of .docx, .odt, .txt, .pdf. Photos of handwritten or printed papers can be uploaded as photos in .jpg, or .png, or other suitable files. Please make sure that your name as a student is on the uploaded document.
The instructor will set the deadline for the assignment. Within the deadline of the assignment multiple attempts are allowed to upload files or photos.
-
Dear students,
The quiz aligns with the following module learning objectives and course objectives/outcomes:
- Obj. 1 Remember advantages of rapid diagnostic testing (C.O. #4: Identify unknown bacteria using various physiological tests.) & (C.O. # 6 : In a virtual setting) or (Face-to-Face setting) Perform essential microbiological techniques in a simulated environment with confidence, utilizing digital tools to analyze and interpret virtual and hands-on experimental data, and apply their virtual and/or hands-on laboratory skills to clinical and public health contexts in nursing and allied health.)
- Obj. 2 Distinguish between different uses of rapid diagnostic tests (C.O. # 4 & C.O. # 6)
- Obj. 3 Distinguish between the different working principles of different rapid diagnostic tests (C.O. # 4 & C.O. # 6).
- Obj. 4 Identify positive, negative, and erroneous or inclusive test result for lateral flow immunochromatography (C.O. # 4 & C.O. # 6)
- Obj. 5 Distinguish between the different working principles of different rapid diagnostic tests (C.O. # 4 & C.O. # 6)
Please take quiz/assignment during dates of availability:
Dates of availability: xx/xx/202x to xx/xx/202x
Attempts: unlimited within dates of availability.
Grade/score: Highest score of attempt counts.
Length of time per attempt: You have xxx minutes for each attempt.
Uninterrupted attempts required.
Number of questions: There are 20 questions.
Contribution towards final grade: yes/no
The quiz has to be taken in the learning management system, in this case Canvas or Moodle.
Email, paper, or any other type of submissions will not be accepted.
Sincerely,
- Obj. 1 Remember advantages of rapid diagnostic testing (C.O. #4: Identify unknown bacteria using various physiological tests.) & (C.O. # 6 : In a virtual setting) or (Face-to-Face setting) Perform essential microbiological techniques in a simulated environment with confidence, utilizing digital tools to analyze and interpret virtual and hands-on experimental data, and apply their virtual and/or hands-on laboratory skills to clinical and public health contexts in nursing and allied health.)
- Obj. 1 Remember advantages of rapid diagnostic testing (C.O. #4: Identify unknown bacteria using various
physiological tests.) & (C.O. # 6 : In a virtual setting) or (Face-to-Face
setting) Perform essential microbiological techniques in a simulated
environment with confidence, utilizing digital tools to analyze and interpret
virtual and hands-on experimental data, and apply their virtual and/or hands-on
laboratory skills to clinical and public health contexts in nursing and allied
health.)
-
Although the historical origins of epidemiology as a science are investigations of epidemics of infectious disease, modern epidemiology has expanded to not only include contagious diseases, but also environmental connections to disease states and even accidental injuries. Epidemiologists gather data on the frequency of various diseases in populations, and correlate risk factors associated with disease development. The information compiled by epidemiologists provides the foundation for the concept of “public health.” (Image: 11098, CDC/ Dr. Terrence Tumpey, CDC Public Health Image Library, Public Domain).
Upon completion of this module, you will be able to:
- Define Epidemiology
- Identify disease patterns and risk factors
- Describe the impact on public health
This module supports Course Outcomes 1, 4, and 5.
To achieve these objectives:
- Read the Module 15 Introduction
- Read Chapter 15 in Medical Microbiology for Nursing and Allied Health Lab Manual
- Review the Epidemiology Lab PowerPoint Slides
- Complete the Module 15 Discussion Forum
- Complete the Epidemiology Quiz
Module Pressbooks Resources and Activities
You will find the following resources and activities in this module at the Pressbooks website. Click on the links below to access or complete each item.
- Define Epidemiology
Background Colour
Font Face
Font Kerning
Font Size
Image Visibility
Letter Spacing
Line Height
Link Highlight
Text Colour